Molecular pathological analysis in routine diagnostic
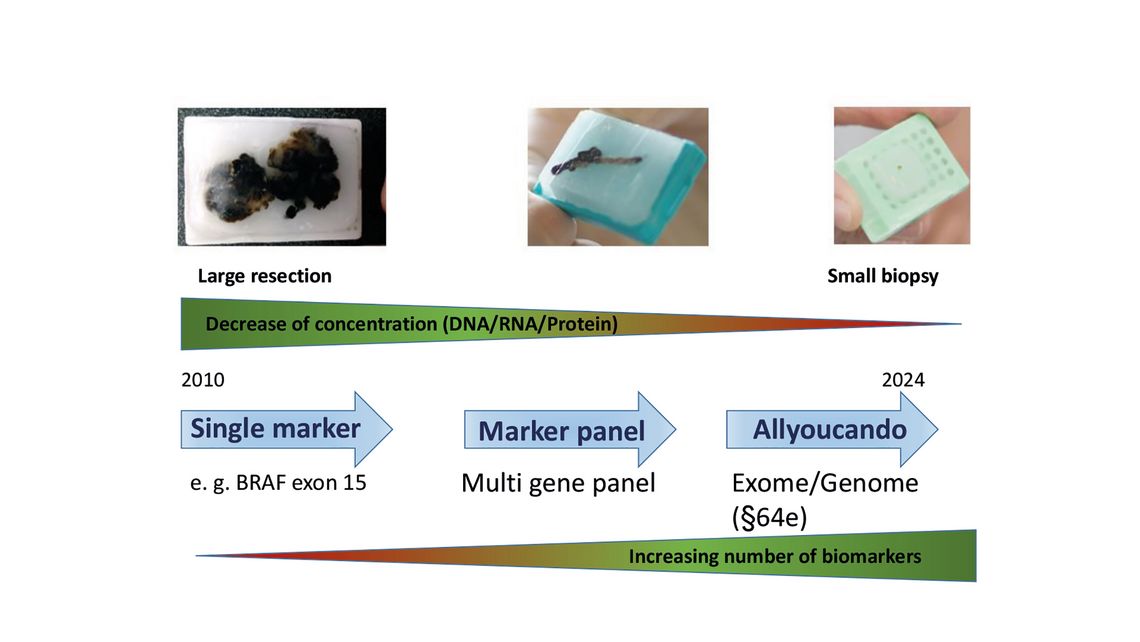
DOI: https://doi.org/10.47184/tp.2023.01.06Molecular pathological examinations in the context of diagnostic analyses of tumour samples cover a broad spectrum of techniques for routine diagnostics. Especially in the last few years, there has been an increase in a large number of new biomarkers, the investigation of which is of great importance for therapy decision but also for accurate diagnosis. At the same time, the available amount of tissue is getting more and more limited due to different reasons: 1. The change in surgical procedure from large resections to tiny little biopsies; 2. The increasing number of markers which has to be tested on this limited material, e. g. by immunohistochemical stainings.
Keywords: Tumor samples, molecular pathology, testing techniques
Pathological specimens used in molecular pathology
The working material that is generally used in pathologies is formalin-fixed paraffin-embedded tissue. Since a high tumor cell content is very important for molecular analysis as the allelic fraction of variants can be in the range from very low (around 5 %) to very high (100 %), the pathologists mark the tumor areas on hematoxilin-eosin stained slides and estimate the tumor cell content in these areas. Nucleic acids, DNA as well as RNA, can both be isolated from these tissues after deparaffinization by macro-dissection of the tumor area(s) from unstained sections of tissue slides and used for a diverse spectrum of techniques. The increase in biomarkers being tested presents new challenges to molecular pathology as the available material becomes more limited. Figure 1 depicts the change in the need for biomarker testing compared to tissue availability and thus available nucleic acids.
Further limitations, which have to be considered, are fixation artifacts in the FFPE material (fixation artefacts due to formalin treatment) and the age of the material, since over the years degrade more and more. Therefore, besides quantity measurements, quality control of the extracted nucleic acids are becoming are also very important.
Spectrum of molecular techniques in Molecular Pathology
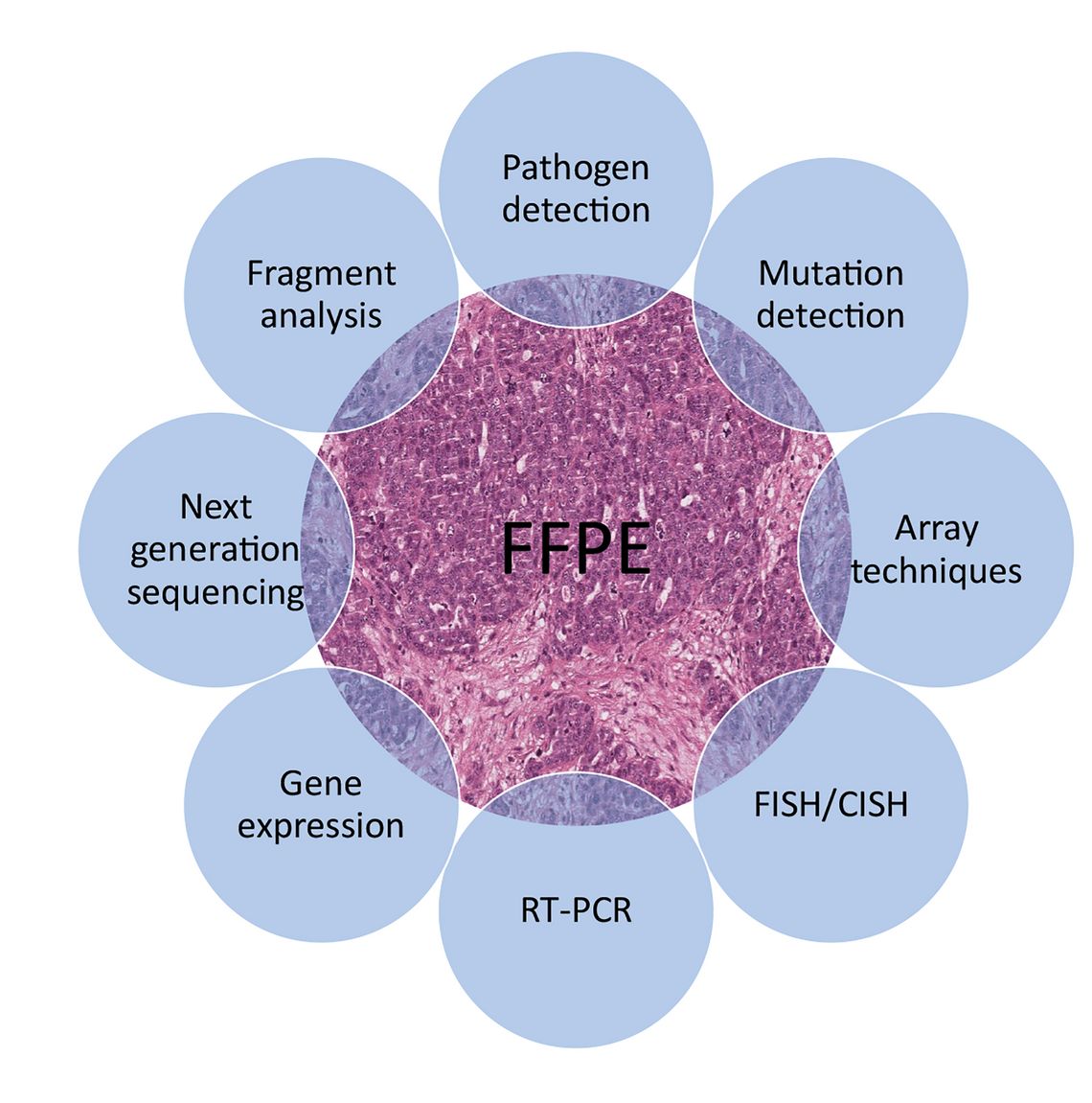
The spectrum of molecular pathological examinations includes so called “classical” detection methods like pathogen detection, fragment length analyses (e. g. microsatellite instability, clonality analyses). But also PCR-based techniques such as mutation-specific (allele-specific) PCR for the specific detection of point mutations at specific positions in genes (absence/presence testing, e. g. BRAF V600E mutation detection), melting curve anaysis (e. g. BRAF V600E mutation detection) and many more (see Figure 2) are commonly used in molecular pathology. Furthermore, the use of more complex techniques like Next Generation Sequencing (NGS) either applying small to large gene panels or Whole Exome Sequencing (WES) and hybridization of arrays are becoming more and more important for routine diagnostics of tumor specimens.
The most common problem in the pathologies is the degradation of the nucleic acids in the FFPE tissue, which is becoming more pronounced every year. As a result, molecular pathology has been forced to develop and use methods to overcome this phenomenon. Therefore, most PCR or NGS-based techniques rely on the use of short fragments than full-length sequencing techniques. However, here too, more and more possibilities are emerging to enhance the capability to analyze even highly degraded tumor samples. Due to the current developments resulting from the model project § 64e SGB V, efforts are increasing to implement whole exome sequencing in pathology.
Besides analysis on DNA-level RNA-based sequencing techniques for detection of gene fusions or expression analysis is getting more and more in the focus of diagnostics. This is necessary due to the development of more and more targeted therapies for specific translocations, such as fusions of the NTRK genes, NRG1 fusions, FGFR fusions among others. Detection of fusions using RNA techniques is based on classical cDNA synthesis. Different techniques can be used in the following steps by classical RT-PCR with gene-specific primers (usable with known fusion partners) and, if necessary, subsequent Sanger sequencing. Alternatively, NGS-based techniques such as anchored multiplex PCR (e. g. Archer FusionPlex assays), amplicon-based sequencing using multiplex PCR or hybrid capture based techniques with enrichment of the regions to be analyzed.
The use of array-based techniques like Infinium EPIC 850K Methylation array from Illumina are recommended by the current guidelines for diagnosis/classification of brain tumors. Other SNP-based arrays (e. g. OncoScan, Infinium CytoSNP 850K) might be useful for the determination of the genomic instability in tumors, e. g. ovarian cancer, caused by defects in the homologous recombination repair which are suitable for therapy with PARP inhibitors but can also be used to analyze the samples for the occurrence of gene amplifications and/or deletions which might also have an impact on therapy decision.
Further developments in molecular pathology
The ever-growing field of new therapy options practically forces the use of high-throughput analysis capabilities, especially with regard to the steadily decreasing availability of tissues. Furthermore, many pathologies (especially at university) lack suitable personnel with experience in performing the mentioned techniques or evaluating the results, especially the availability of technical staff is a constant problem. In addition, it requires medical staff to have knowledge that goes beyond the knowledge taught in their studies or working field. This therefore creates a new field of activity in the medical sector, as more and more natural scientists are taking over those tasks in the laboratories in cooperation with pathologists.
Conclusions
Diagnostics in the molecular pathology field is challenging in many ways. On the one hand, the challenges are caused by the increase in biomarkers to be tested for both diagnosis and therapy decisions. On the other hand, the decreasing availability of tumor material with which to perform the analyses. This is compounded by the lack of personnel with training in molecular pathology. On the other hand, a new field of activity for natural scientists has been emerging for some years now, who are increasingly entering the field of medical care and performing these diagnostics in collaboration with pathologists.